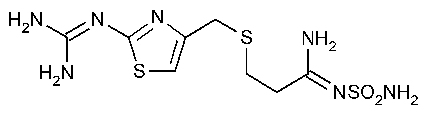
Famotidine
Propanimidamide,N¢-(aminosulfonyl)-3-[[[2-[(diaminomethylene)amino]-4-thiazolyl]methyl]thio]-.
[1-Amino-3-[[[2-[(diaminomethylene)amino]-4-thiazolyl]methyl]thio]propylidene]sulfamide [76824-35-6].
»Famotidine contains not less than 98.5percent and not more than 101.0percent of C8H15N7O2S3,calculated on the dried basis.
Packaging and storage—
Preserve in well-closed containers,protected from light.
Identification—
A:
Infrared Absorption á197Kñ.
B:
Ultraviolet Absorption á197Uñ—
Solution:
25µg per mL.
Medium:
phosphate buffer.
Absorptivities at 265nm,calculated on the dried basis,do not differ by more than 3.0%.[NOTE—Prepare the phosphate buffer as follows.Adjust 250mLof 0.02Mphosphoric acid with sodium hydroxide solution (1in 10)to a pHof 2.5,dilute with water to 500mL,and mix.]
Loss on drying á731ñ—
Dry it at a pressure between 1and 5mm of mercury at 80 for 5hours:it loses not more than 0.5%of its weight.
for 5hours:it loses not more than 0.5%of its weight.
Residue on ignition á281ñ:
not more than 0.1%.
Heavy metals,Method IIá231ñ:
not more than 0.001%.
Chromatographic purity—
Adsorbent:
0.25-mm layer of chromatographic silica gel mixture.
Test solution—
Transfer about 200mg of Famotidine,accurately weighed,to a 10-mLvolumetric flask,add 2mLof methanol,and shake for 10minutes.Add 0.1mLof glacial acetic acid,stir until dissolved,dilute with methanol to volume,and mix.
Standard solutions—
Dissolve an accurately weighed portion of USP Famotidine RSin a mixture of methanol and glacial acetic acid (100:1)to obtain Standard solution 1having a known concentration of 0.2mg per mL.Dilute a portion of this solution,accurately measured,with a mixture of methanol and glacial acetic acid (100:1)to obtain Standard solution 2containing 65µg of USP Famotidine RSper mL.
Developing solvent system:
a mixture of ethyl acetate,methanol,toluene,and ammonium hydroxide (40:25:20:2).
Procedure—
Separately apply 5µLof the Test solutionand 5µLof each Standard solutionto a plate,and dry under a stream of nitrogen.Proceed as directed for Thin-Layer Chromatographyunder Chromatography á621ñ.Examine the plate under short-wavelength UVlight,and compare the intensities of any secondary spots observed in the chromatogram of the Test solutionwith those of the principal spots in the chromatograms of the Standard solutions:no secondary spot from the chromatogram of the Test solutionis larger in size or more intense than the principal spot obtained from Standard solution 2(0.3%);and the sum of the intensities of the secondary spots obtained from the Test solutioncorresponds to not more than 1.0%(Standard solution 1).
Organic volatile impurities,Method IVá467ñ:
meets the requirements.
Solvent—
Use dimethyl sulfoxide.
Assay—
Dissolve about 250mg of Famotidine,accurately weighed,in 80mLof glacial acetic acid,and titrate with 0.1Nperchloric acid VS(see Titrimetry á541ñ),using a suitable anhydrous electrode system.Any aqueous electrolyte solution contained in the electrodes employed should be removed,the electrode rendered anhydrous and filled with 0.1Nlithium perchlorate in acetic anhydride.Perform a blank determination and make any necessary correction.Each mLof 0.1Nperchloric acid is equivalent to 16.87mg of C8H15N7O2S3.
Auxiliary Information—
Staff Liaison:Elena Gonikberg,Ph.D.,Scientist
Expert Committee:(PA4)Pharmaceutical Analysis 4
USP28–NF23Page 805
Phone Number:1-301-816-8251