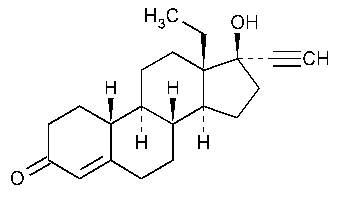
Norgestrel
18,19-Dinorpregn-4-en-20-yn-3-one,13-ethyl-17-hydroxy-,(17a)-(±)-.
(±)-13-Ethyl-17-hydroxy-18,19-dinor-17a-pregn-4-en-20-yn-3-one [6533-00-2].
»Norgestrel contains not less than 98.0percent and not more than 102.0percent of C21H28O2,calculated on the dried basis.
Packaging and storage—
Preserve in well-closed containers.
Identification,Infrared Absorption á197Kñ—
If differences appear,dissolve portions of both the test specimen and the Reference Standard in ethyl acetate,evaporate the solutions on a steam bath to dryness,and repeat the test.
Melting range,Class Iá741ñ:
between 205 and 212
and 212 ,but the range between beginning and end of melting does not exceed 4
,but the range between beginning and end of melting does not exceed 4 .
.
Optical rotation á781Añ:
between -0.1 and +0.1.
and +0.1.
Test solution:
50mg,previously dried,per mL,in chloroform.
Loss on drying á731ñ—
Dry it at 105 for 3hours:it loses not more than 0.5%of its weight.
for 3hours:it loses not more than 0.5%of its weight.
Residue on ignition á281ñ:
not more than 0.3%.
Chromatographic purity—
Phosphomolybdic acid reagent—
Add 10g of phosphomolybdic acid to 100mLof alcohol,and stir the mixture for not less than 30minutes.Filter before use.
Test preparation—
Prepare a solution of Norgestrel in chloroform to contain 10.0mg per mL.
Standard solution andStandard dilutions—
Prepare a solution of USP Norgestrel RSin chloroform to contain 10mg per mL(Standard solution).Prepare a series of dilutions of Standard solutionin chloroform to contain 0.20,0.10,0.05,0.02,and 0.01mg per mL(Standard dilutions).
Procedure—
Apply 10-µLvolumes of Standard solution,the Test preparation,and each of the five Standard dilutionsat equidistant points along a line 2.5cm from one edge of a 20-×20-cm thin-layer chromatographic plate (see Chromatography á621ñ)coated with a 0.25-mm layer of chromatographic silica gel mixture and previously activated by heating at 100 for 15minutes.Place the plate in a suitable developing chamber that contains and has been equilibrated with a mixture of 96volumes of chloroform and 4volumes of alcohol,seal the chamber,and allow the chromatogram to develop until the solvent front has moved 15cm above the line of application.Remove the plate,allow the solvent to evaporate,then spray uniformly with Phosphomolybdic acid reagent,and heat it at 105
for 15minutes.Place the plate in a suitable developing chamber that contains and has been equilibrated with a mixture of 96volumes of chloroform and 4volumes of alcohol,seal the chamber,and allow the chromatogram to develop until the solvent front has moved 15cm above the line of application.Remove the plate,allow the solvent to evaporate,then spray uniformly with Phosphomolybdic acid reagent,and heat it at 105 for 10to 15minutes.The lane of the Test preparationexhibits its principal spot at the same RFas the principal spot of Standard solution.If spots other than the principal spot are observed in the lane of the Test preparation,estimate the concentration of each by comparison with the Standard dilutions.The spots from the 0.20-,0.10-,0.05-,0.02-,and 0.01-mg per mLdilutions are equivalent to 2.0,1.0,0.5,0.2,and 0.1%of impurities,respectively.The requirements of the test are met if the sum of the impurities in the Test preparationdoes not exceed 2.0%.
for 10to 15minutes.The lane of the Test preparationexhibits its principal spot at the same RFas the principal spot of Standard solution.If spots other than the principal spot are observed in the lane of the Test preparation,estimate the concentration of each by comparison with the Standard dilutions.The spots from the 0.20-,0.10-,0.05-,0.02-,and 0.01-mg per mLdilutions are equivalent to 2.0,1.0,0.5,0.2,and 0.1%of impurities,respectively.The requirements of the test are met if the sum of the impurities in the Test preparationdoes not exceed 2.0%.
Limit of ethynyl group—
Proceed as directed in the test for Ethynyl groupunder Norethindrone.Not less than 7.81%and not more than 8.18%of ethynyl group is found.
Assay—
Dissolve about 100mg of Norgestrel,accurately weighed,in alcohol,and dilute quantitatively and stepwise with alcohol to obtain a solution containing about 10µg per mL.Dissolve an accurately weighed quantity of USP Norgestrel RSin alcohol to obtain a Standard solution having a known concentration of about 10µg per mL.Concomitantly determine the absorbances of both solutions in 1-cm cells at the wavelength of maximum absorbance at about 241nm,with a suitable spectrophotometer,using alcohol as the blank.Calculate the quantity,in mg,of C21H28O2in the portion of Norgestrel taken by the formula:
10C(AU/AS),
in which Cis the concentration,in µg per mL,of USP Norgestrel RSin the Standard solution,and AUand ASare the absorbances of the solution of Norgestrel and the Standard solution,respectively.
Auxiliary Information—
Staff Liaison:Clydewyn M.Anthony,Ph.D.,Scientist
Expert Committee:(PA1)Pharmaceutical Analysis 1
USP28–NF23Page 1402
Phone Number:1-301-816-8139