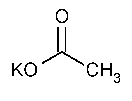
Potassium Acetate
»Potassium Acetate contains not less than 99.0percent and not more than 100.5percent of C2H3KO2,calculated on the dried basis.
Packaging and storage—
Preserve in tight containers.
Identification—
Asolution (1in 10)responds to the tests for Potassium á191ñand for Acetate á191ñ.
pHá791ñ:
between 7.5and 8.5,in a solution (1in 20).
Loss on drying á731ñ—
Dry it at 150 for 2hours:it loses not more than 1.0%of its weight.
for 2hours:it loses not more than 1.0%of its weight.
Heavy metals,Method Iá231ñ—
Prepare the Test Preparationas follows.Dissolve 1g in 10mLof water,add 3.0mLof glacial acetic acid,dilute with water to 25mL,and adjust with glacial acetic acid to a pHbetween 3.8and 4.0,measured with a pHmeter.Prepare the Monitor Preparationas directed for Test Preparation,2.0mLof Standard Lead Solutionbeing added:the limit is 0.002%.
Limit of sodium—
Potassium chloride solution—
Dissolve 100g of potassium chloride in water to make 1000mL.
Standard solutions—
Transfer 127.1mg of sodium chloride,previously dried at 105 for 2hours and accurately weighed,to a 500-mLvolumetric flask,add water to volume,and mix.Transfer 10.0mLof this solution to a 100-mLvolumetric flask,dilute with water to volume,and mix.Transfer 2.0,5.0,and 10.0mLof this solution to separate 100-mLvolumetric flasks,add 10.0mLof Potassium chloride solutionto each flask,dilute with water to volume,and mix.These Standard solutionscontain 0.2,0.5,and 1.0µg of sodium per mL,respectively.[NOTE—Concentrations of sodium in the Standard solutions may be modified to fit the linear or working range of the atomic absorption spectrophotometer.]
for 2hours and accurately weighed,to a 500-mLvolumetric flask,add water to volume,and mix.Transfer 10.0mLof this solution to a 100-mLvolumetric flask,dilute with water to volume,and mix.Transfer 2.0,5.0,and 10.0mLof this solution to separate 100-mLvolumetric flasks,add 10.0mLof Potassium chloride solutionto each flask,dilute with water to volume,and mix.These Standard solutionscontain 0.2,0.5,and 1.0µg of sodium per mL,respectively.[NOTE—Concentrations of sodium in the Standard solutions may be modified to fit the linear or working range of the atomic absorption spectrophotometer.]
Test solution—
Transfer about 0.2g of Potassium Acetate,accurately weighed,to a 100-mLvolumetric flask containing about 50mLof water,and swirl to dissolve.Add 10.0mLof Potassium chloride solution,dilute with water to volume,and mix.[NOTE—The concentration of Potassium Acetate in the Test solution may be modified by using a different quantity or by further dilution to bring the absorption response within the range of responses obtained from the Standard solutions.]
Blank solution—
Transfer 10.0mLof Potassium chloride solutionto a 100-mLvolumetric flask,dilute with water to volume,and mix.
Procedure—
Concomitantly determine the absorbances of the Standard solutionsand the Test solutionat the sodium emission line of 589nm,with a suitable atomic absorption spectrophotometer (see Spectrophotometry and Light-Scattering á851ñ)equipped with a sodium hollow-cathode lamp and an oxidizing air–acetylene flame,using the Blank solutionto zero the instrument.Plot the absorbances of the Standard solutionsversus concentration,in µg per mL,of sodium,and draw the straight line best fitting the plotted points.From the graph so obtained,determine the concentration C,in µg per mL,of sodium in the Test solution.Calculate the percentage of sodium in the portion of Potassium Acetate taken by the formula:
CD/10,000W,
in which Wis the quantity,in g,of Potassium Acetate taken to prepare the Test solution;and Dis the extent of dilution of the Test solution:not more than 0.03%of sodium is found.
Assay—
Dissolve about 200mg of Potassium Acetate,previously dried and accurately weighed,in 25mLof glacial acetic acid,add 2drops of crystal violet TS,and titrate with 0.1Nperchloric acid VSto a green endpoint.Perform a blank determination,and make any necessary correction.Each mLof 0.1Nperchloric acid is equivalent to 9.814mg of C2H3KO2.
Auxiliary Information—
Staff Liaison:Lawrence Evans,III,Ph.D.,Scientist
Expert Committee:(PA6)Pharmaceutical Analysis 6
USP28–NF23Page 1580
Pharmacopeial Forum:Volume No.28(4)Page 1185
Phone Number:1-301-816-8389