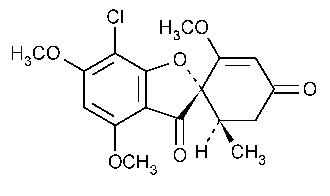
Griseofulvin
Spiro[benzofuran-[2](3H),1¢-2cyclohexene]-3,4¢-dione,7-chloro-2¢,4,6-trimethoxy-6¢-methyl-,(1¢S-trans)-.
7-Chloro-2¢,4,6-trimethoxy-6¢b-methylspiro[benzofuran-2(3H),1¢-[2]cyclohexene]-3,4¢-dione [126-07-8].
»Griseofulvin has a potency of not less than 900µg of C17H17ClO6per mg.
Packaging and storage—
Preserve in tight containers.
Identification—
A:
Infrared Absorption á197Mñ.
B:
The retention time of the major peak in the chromatogram of the Assay preparationcorresponds to that in the chromatogram of the Standard preparation,as obtained in the Assay.
Specific rotation á781Sñ:
between +348 and +364
and +364 .
.
Test solution:
10mg per mL,in dimethylformamide.
Crystallinity á695ñ:
meets the requirements.
Loss on drying á731ñ—
Dry about 100mg,accurately weighed,in a capillary-stoppered bottle in vacuum at a pressure not exceeding 5mm of mercury at 60 for 3hours:it loses not more than 1.0%of its weight.
for 3hours:it loses not more than 1.0%of its weight.
Residue on ignition á281ñ:
not more than 0.2%.
Permeability diameter—
Determine the apparent particle size in µm by the air-permeation method,using a suitable subsieve sizer.Weigh 1.819±0.001g of Griseofulvin,and transfer to the compression tube of the apparatus.Compact with moderate pressure so that a uniform porosity is achieved.Pass dry compressed air through the tube,and measure the air pressure with a water manometer.Read the porosity,and calculate the apparent particle size from the instrument equation.Repeat the porosity readings at successively higher degrees of compaction until the apparent particle size reaches a minimum value.Calculate the observed permeability diameter,in square meters per g,taken by the formula:
Concomitantly determine the observed permeability diameter of a similar preparation of USP Griseofulvin Permeability Diameter RS.Calculate the permeability diameter of the Griseofulvin taken by the formula:
6/(1.455MF),
in whichMis the minimum apparent particle size,and Fis a factor,obtained from the accompanying table,interpolation being used if necessary,to correct the apparent particle size to the true particle size at a given porosity reading.
| Porosity
Reading |
F | Porosity
Reading |
F |
| 0.80 | 1.3771 | 0.56 | 1.7353 |
| 0.76 | 1.4142 | 0.52 | 1.8528 |
| 0.72 | 1.4573 | 0.48 | 2.0076 |
| 0.68 | 1.5082 | 0.44 | 2.2203 |
| 0.64 | 1.5690 | 0.40 | 2.5298 |
| 0.60 | 1.6432 |
OU(AS/OS),
in which OUis the observed permeability diameter of the specimen,ASis the assigned permeability diameter of USP Griseofulvin Permeability Diameter RS,and OSis the observed permeability diameter of USP Griseofulvin Permeability Diameter RS:it is between 1.3and 1.7square meters per g.
Heavy metals,Method IIá231ñ:
0.0025%.
Organic volatile impurities,Method Vá467ñ:
meets the requirements.
Solvent—
Use dimethyl sulfoxide.
Assay—
Mobile phase—
Prepare a suitable filtered mixture of water,acetonitrile,and tetrahydrofuran (60:35:5).Degas for 5minutes before use,and stir continuously during use.Make adjustments if necessary (see System Suitabilityunder Chromatography á621ñ).
Standard preparation—
Dissolve an accurately weighed quantity of USP Griseofulvin RSin methanol to obtain a solution having a known concentration of about 1.25mg per mL.Transfer 5.0mLof this solution to a 50-mLvolumetric flask,dilute with Mobile phaseto volume,and mix.This solution contains about 0.125mg of USP Griseofulvin RSin each mL.
Assay preparation—
Transfer about 62mg of Griseofulvin,accurately weighed,to a 50-mLvolumetric flask,dissolve in and dilute with methanol to volume,and mix.Transfer 5.0mLof this solution to a 50-mLvolumetric flask,dilute with Mobile phaseto volume,and mix.
Chromatographic system
(see Chromatography á621ñ)—The liquid chromatograph is equipped with a 254-nm detector and a 4.6-mm ×25-cm column that contains packing L10.The flow rate is about 1mLper minute.The relative standard deviation for replicate injections of Standard preparationis not more than 2.0%.
Procedure—
Separately inject equal volumes (about 20µL)of the Standard preparationand the Assay preparationinto the chromatograph,and measure the peak responses for the major peaks.Calculate the quantity,in µg of C17H17ClO6,in each mg of the Griseofulvin taken by the formula:
500(CP/WU)(rU/rS),
in which Cis the concentration,in mg per mL,of USP Griseofulvin RSin the Standard preparation;Pis the content,in µg of C17H17ClO6per mg,of USP Griseofulvin RS;WUis the quantity,in mg,of Griseofulvin taken;and rUand rSare the griseofulvin peak responses obtained from the Assay preparationand the Standard preparation,respectively.
Auxiliary Information—
Staff Liaison:William W.Wright,Ph.D.,Scientific Fellow
Expert Committee:(PA7)Pharmaceutical Analysis 7
USP28–NF23Page 920
Phone Number:1-301-816-8335