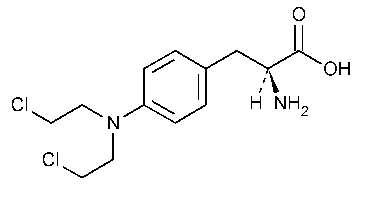
Melphalan
L-Phenylalanine,4-bis(2-chloroethyl)amino]-.
L-3-[p-[Bis(2-chloroethyl)amino]phenyl]alanine [148-82-3].
»Melphalan contains not less than 93.0percent and not more than 100.5percent of C13H18Cl2N2O2,calculated on the dried and ionizable chlorine-free basis.
Packaging and storage—
Preserve in tight,light-resistant,glass containers.
Identification—
B:
To 1mLof 1in 10,000solution in alcohol in a glass-stoppered test tube add 1mLof pH4.0acid phthalate buffer (see under Solutionsin the section Reagents,Indicators,and Solutions),1mLof a 1in 20solution of 4-(p-nitrobenzyl)pyridine in acetone,and 1mLof saline TS.Heat on a water bath at 80 for 20minutes,and cool quickly.Add 10mLof alcohol and 1mLof 1Npotassium hydroxide:a violet to red-violet color is produced.
for 20minutes,and cool quickly.Add 10mLof alcohol and 1mLof 1Npotassium hydroxide:a violet to red-violet color is produced.
C:
Heat 100mg with 10mLof 0.1Nsodium hydroxide on a water bath for 10minutes:the resulting solution,after acidification with 2Nnitric acid,responds to the tests for Chloride á191ñ.
Specific rotation á781Sñ:
between -30 and -36
and -36 .
.
Test solution:
7mg per mL,in methanol,prepared with the aid of gentle heating.
Loss on drying á731ñ:
Dry it in vacuum at 105 to constant weight:it loses not more than 7.0%of its weight.
to constant weight:it loses not more than 7.0%of its weight.
Residue on ignition á281ñ:
not more than 0.3%.
Ionizable chlorine—
Dissolve about 500mg of Melphalan,accurately weighed,in a mixture of 75mLof water and 2mLof nitric acid,allow to stand for 2minutes,and titrate with 0.1Nsilver nitrate VS,determining the endpoint potentiometrically:not more than 1.0mLof 0.1Nsilver nitrate is required for each 500mg of test specimen.
Nitrogen content á461ñ—
Determine the nitrogen content as directed under Method II,using about 325mg of Melphalan,accurately weighed,and 0.1Nsulfuric acid VSfor the titration:not less than 8.90%and not more than 9.45%of Nis found,calculated on the dried basis.
Organic volatile impurities,Method IVá467ñ:
meets the requirements.
Assay—
Transfer to a beaker about 200mg of Melphalan,accurately weighed,and dissolve in 20mLof 0.5Nsodium hydroxide.Cover the beaker with a watch glass,and boil the solution for 30minutes,adding water as necessary to maintain the volume.Cool,neutralize to phenolphthalein TSwith acetic acid,and add 1mLof acetic acid in excess.Titrate with 0.1Nsilver nitrate VS,determining the endpoint potentiometrically,using silver and calomel electrodes,the latter modified to contain saturated potassium sulfate solution.From the results obtained in the test for Ionizable chlorine,calculate the volume,in mL,of 0.1Nsilver nitrate that is equivalent to the ionizable chlorine in the quantity of Melphalan taken for the Assay,and subtract it from the Assaytitration volume.Each mLof 0.1Nsilver nitrate is equivalent to 15.26mg of C13H18Cl2N2O2.
Auxiliary Information—
Staff Liaison:Lawrence Evans,III,Ph.D.,Scientist
Expert Committee:(PA6)Pharmaceutical Analysis 6
USP28–NF23Page 1202
Phone Number:1-301-816-8389